The Nuclear Lamina Is Composed Of Lamin Proteins

Lamin b was one of the first proteins shown to be modified by prenylation 89.
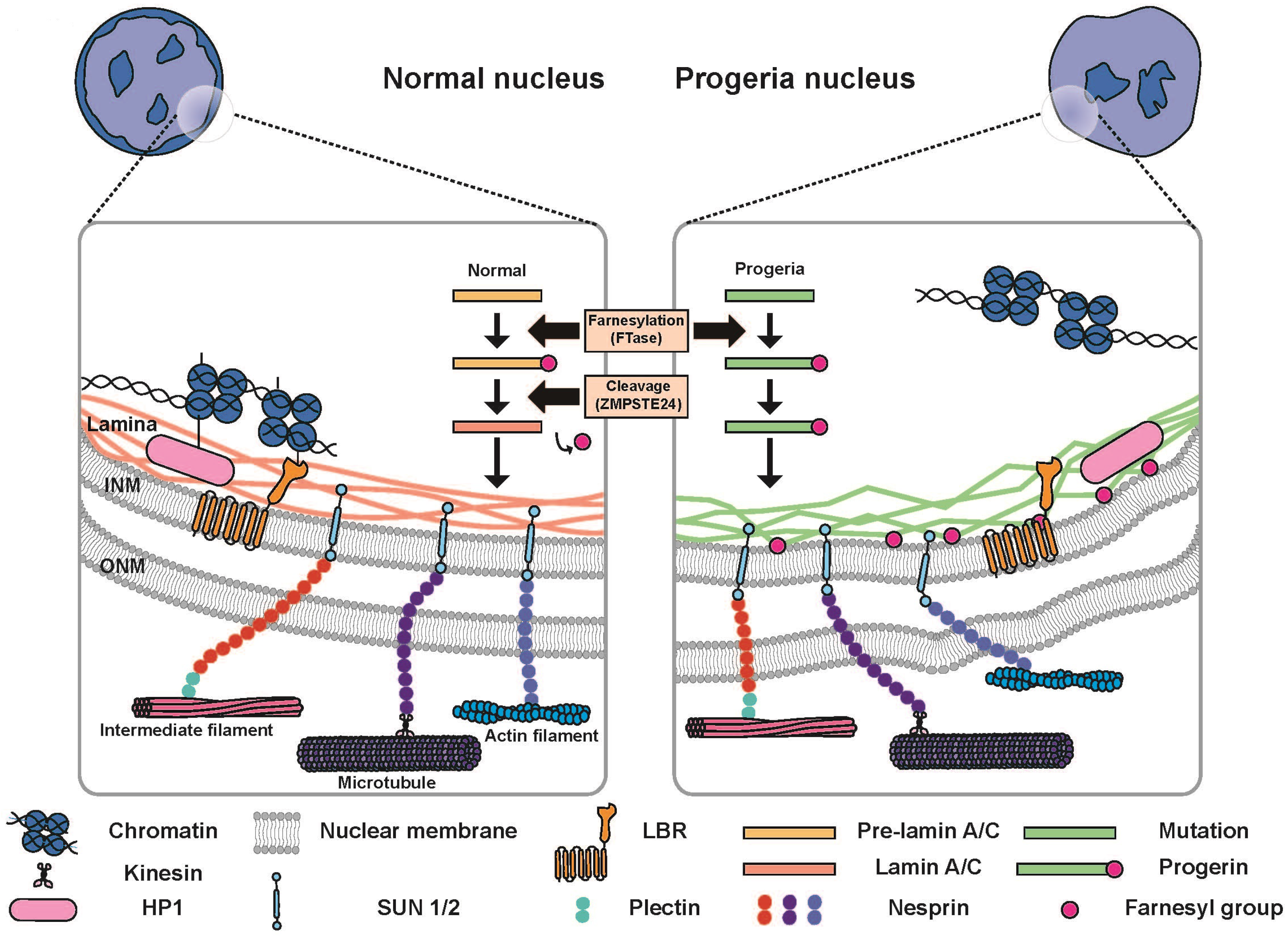
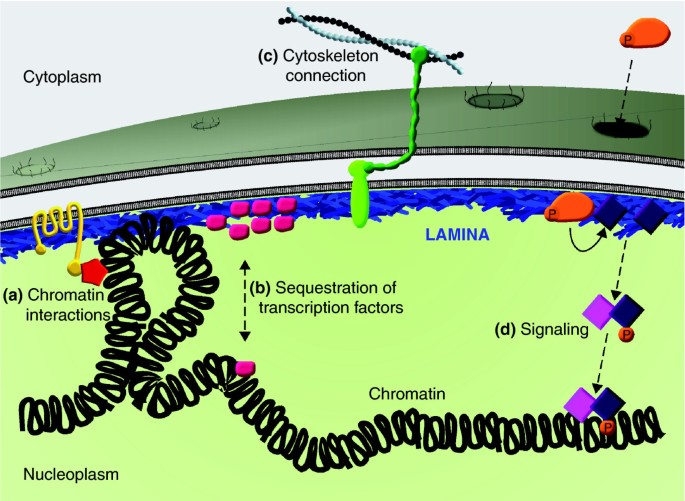
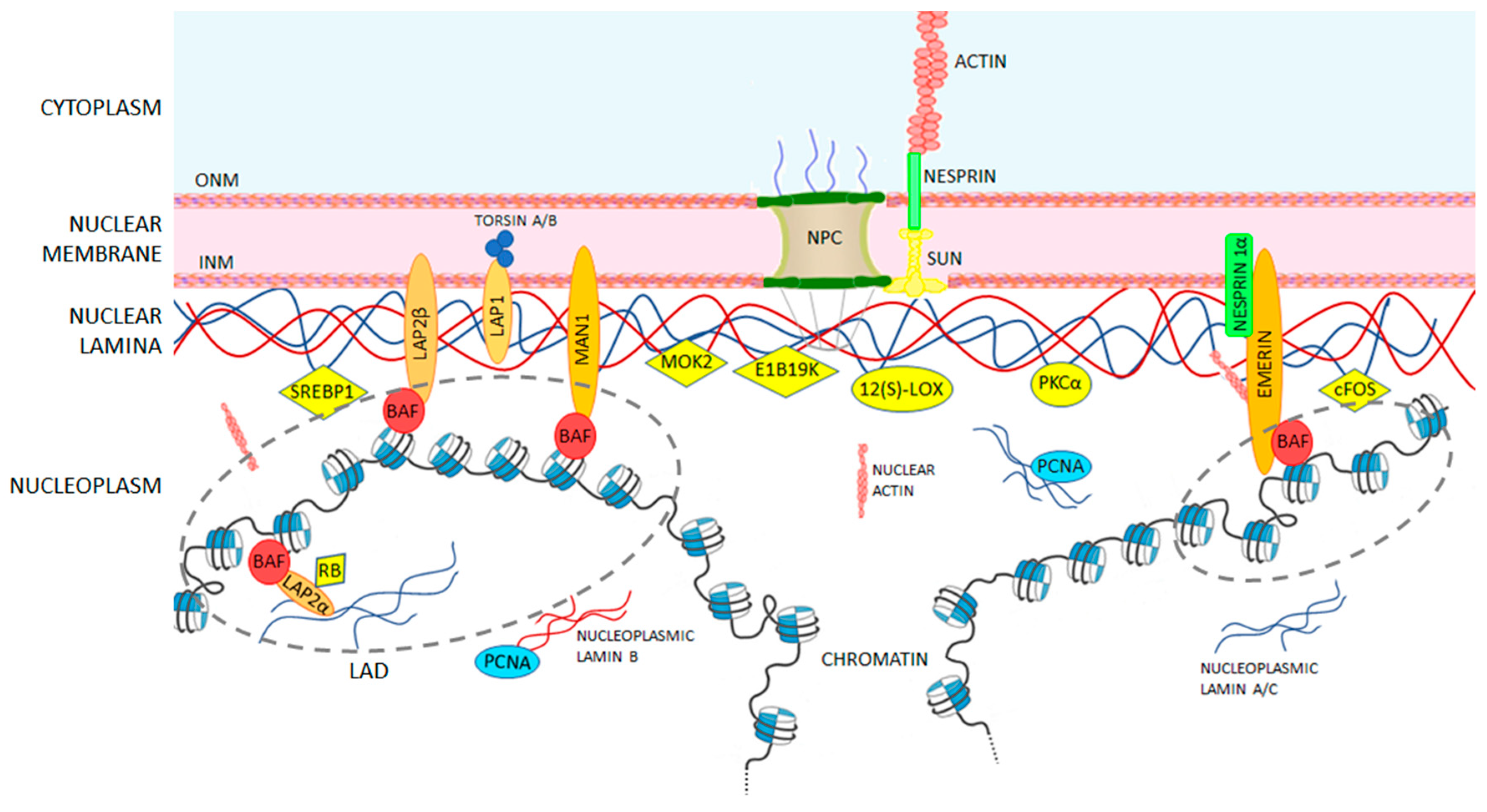
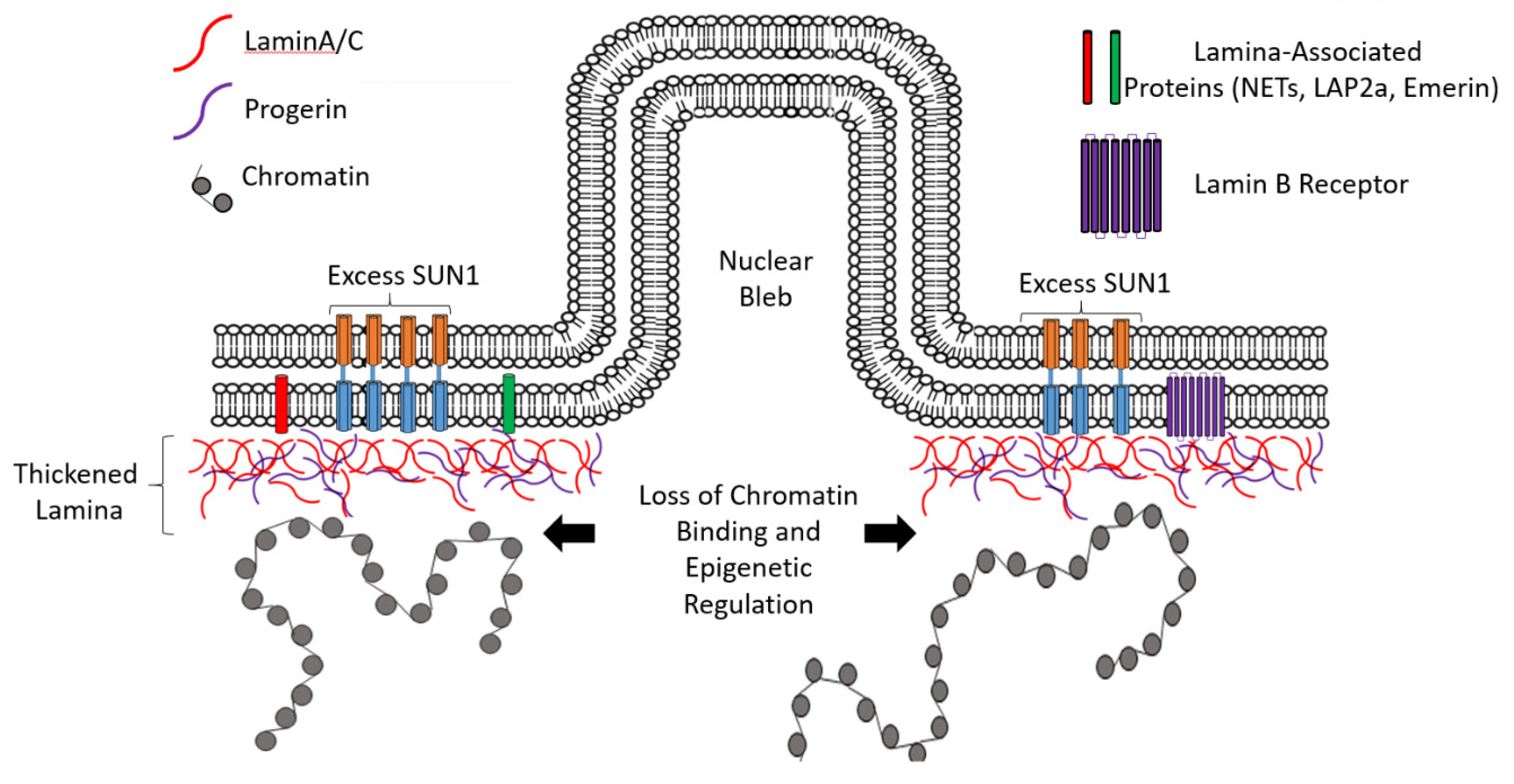
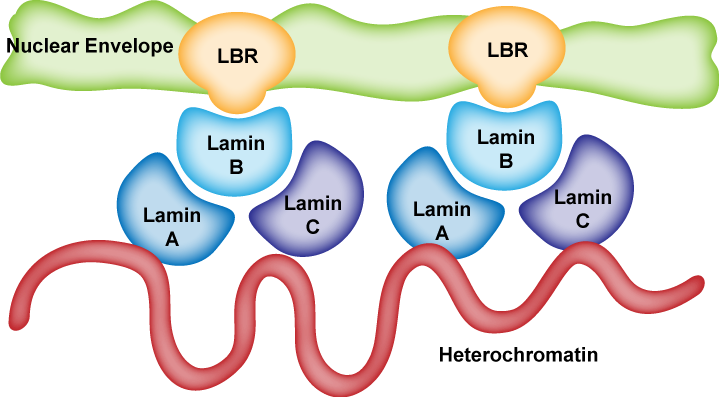
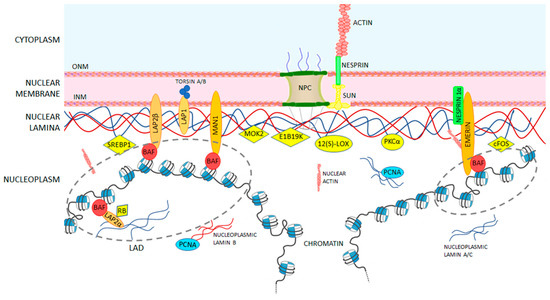
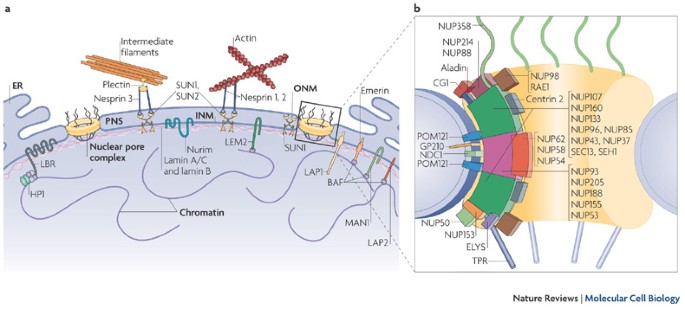
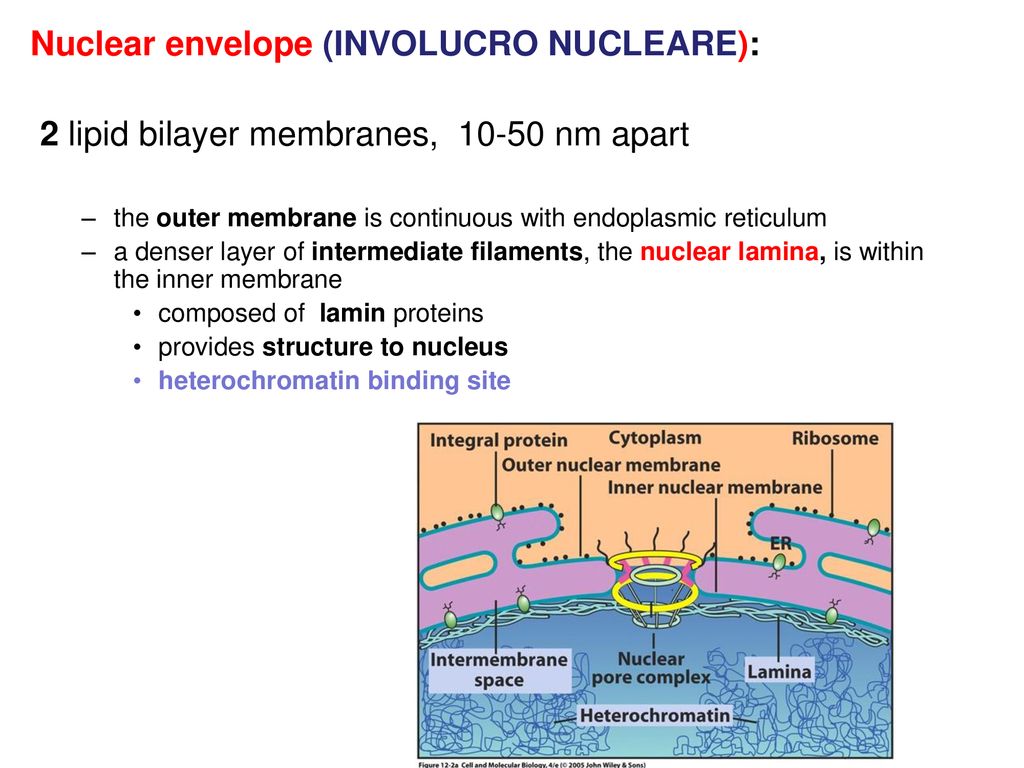
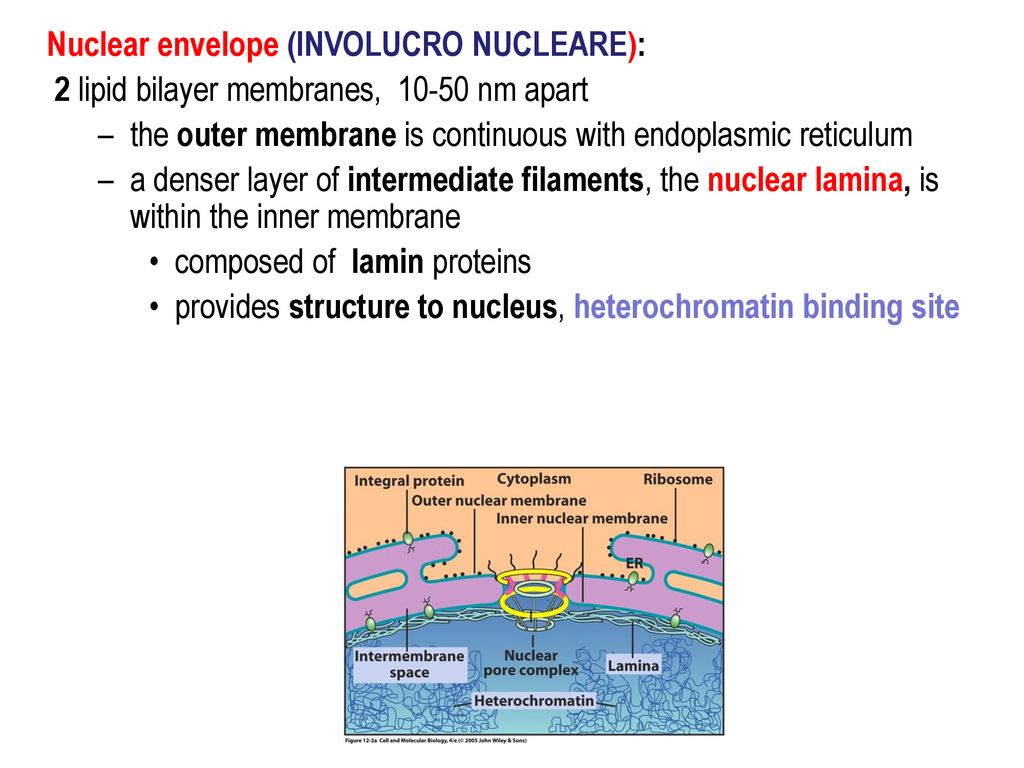
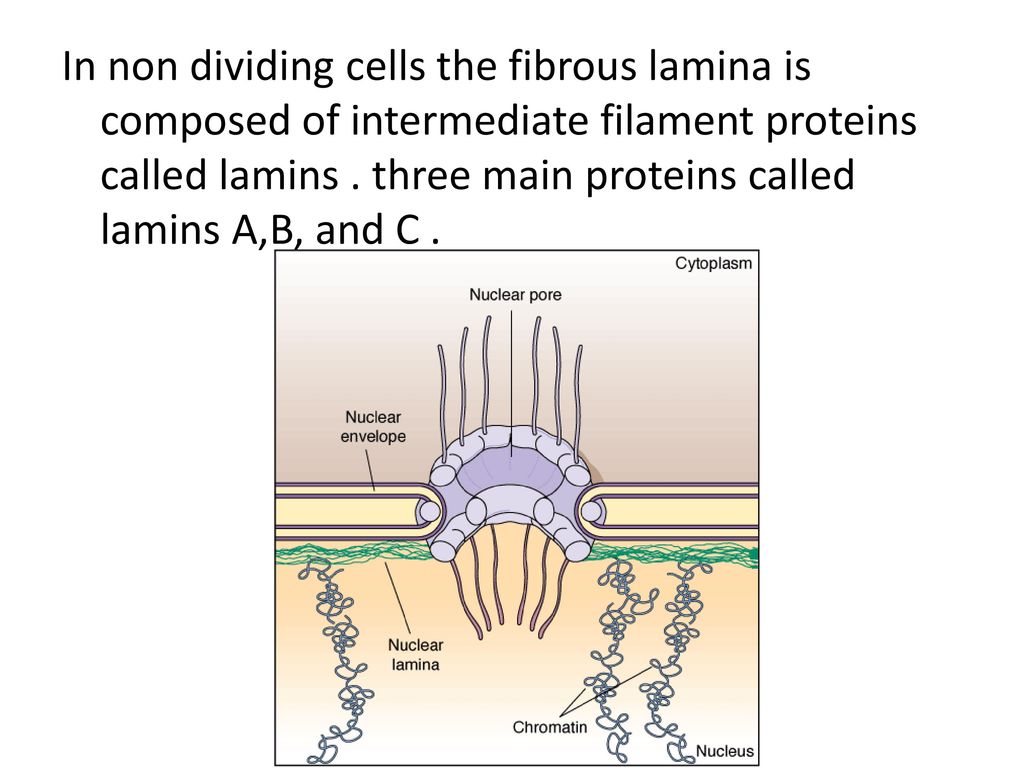
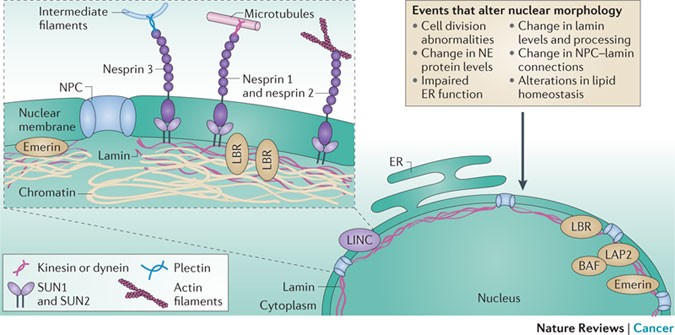
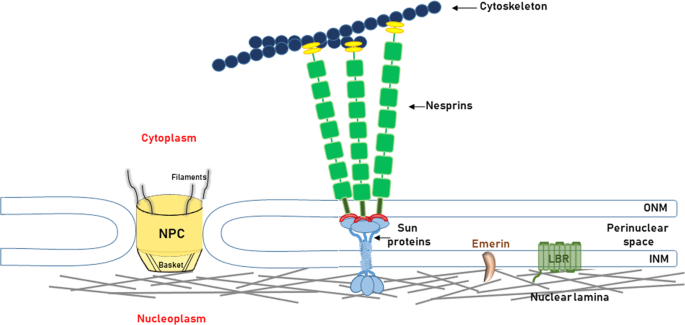
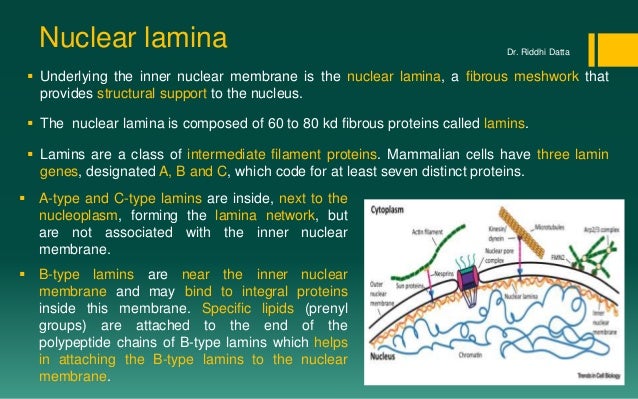
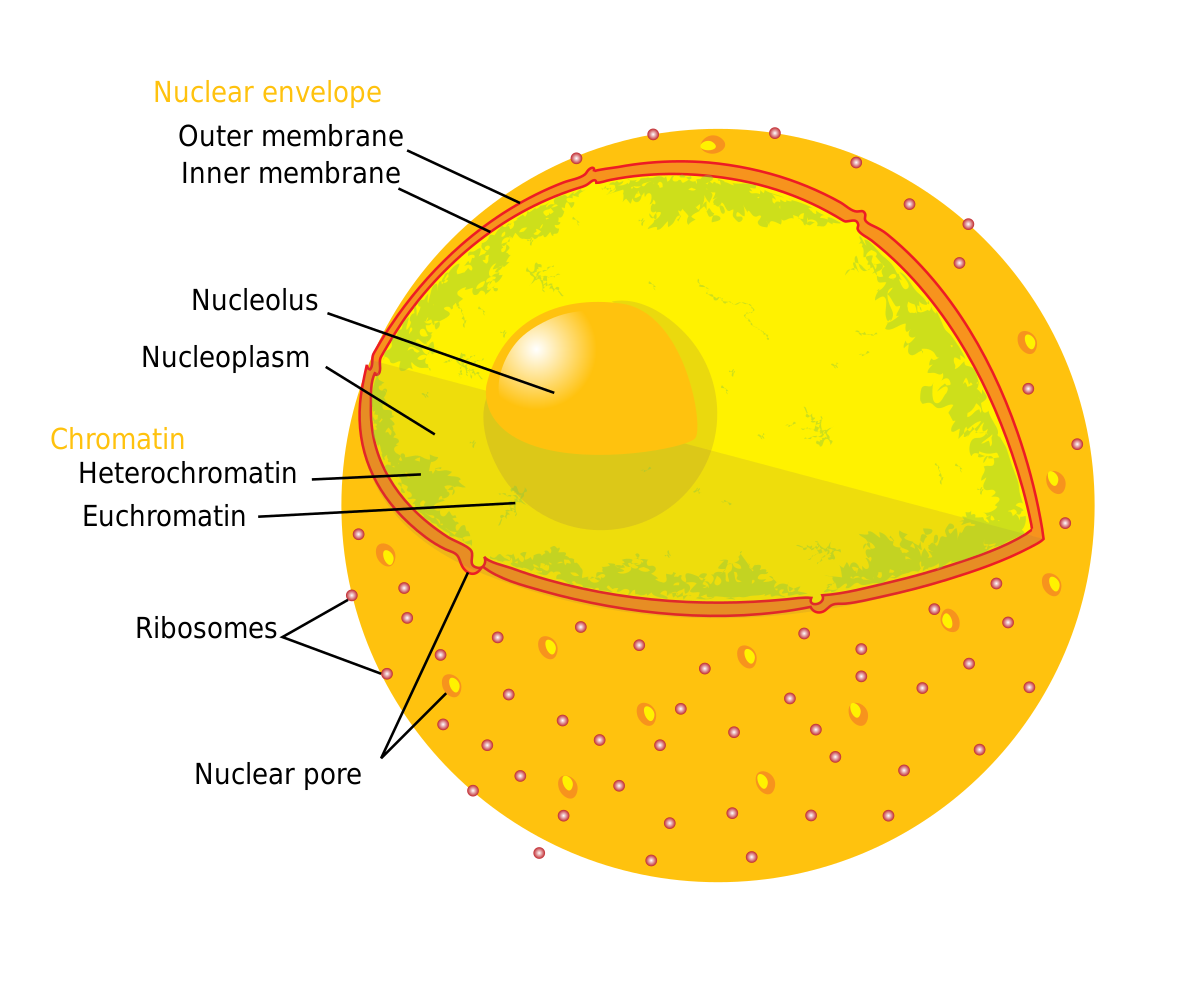
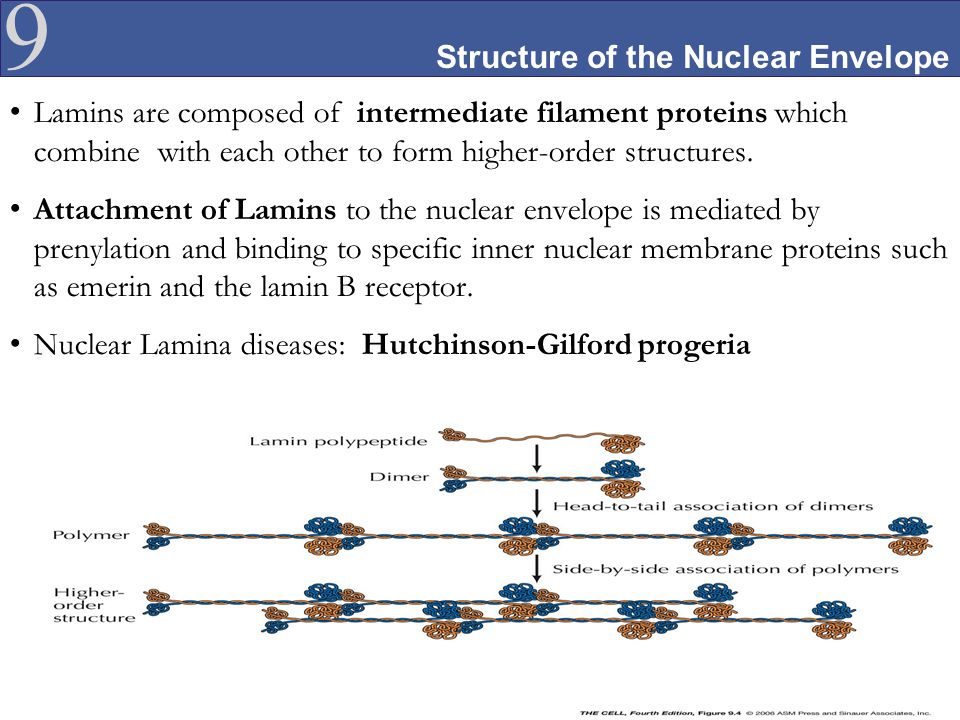
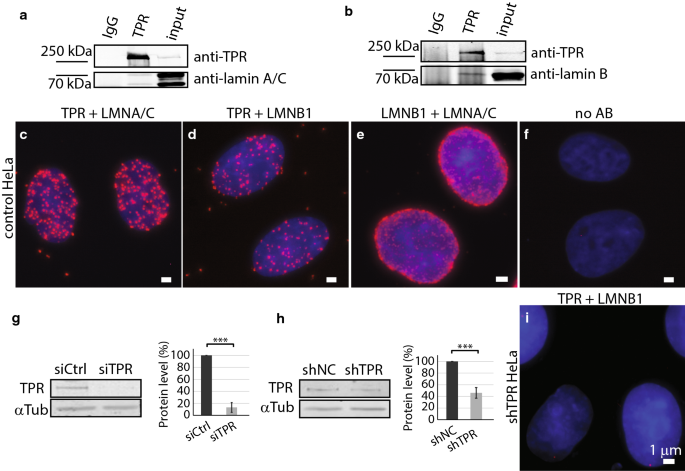
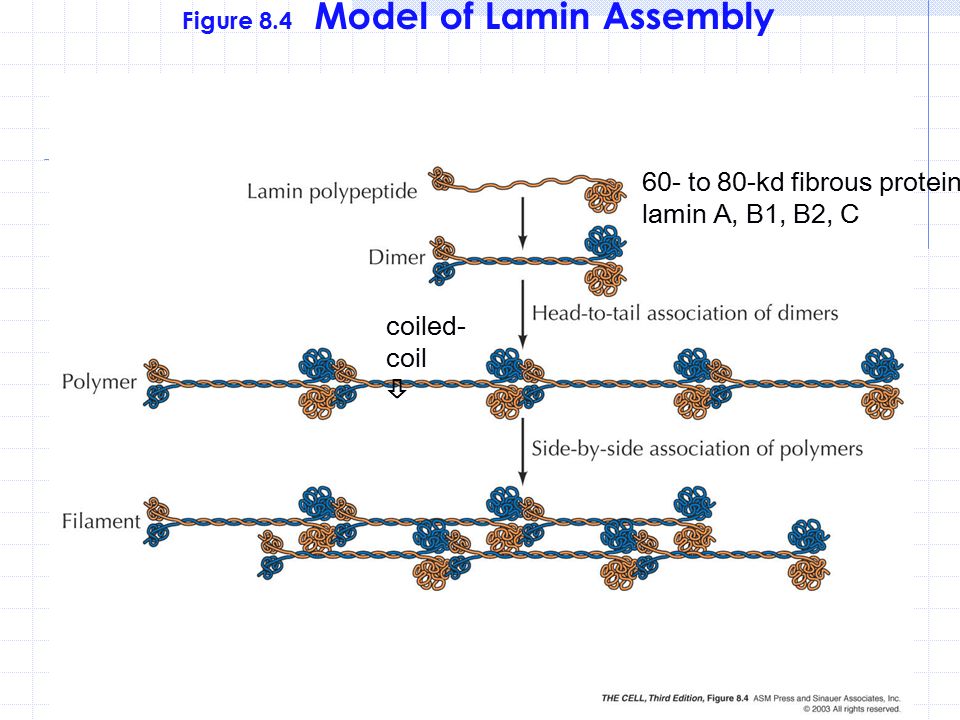
The nuclear lamina is composed of lamin proteins. The nuclear lamina is a multi protein lattice composed of a and b type lamins and their associated proteins. Composed of lamins and lamin associated proteins which include emerin and lbr among others 7 8 the 10 30 nm thick nuclear lamina is found ubiquitously in differentiated cells. This protein lattice associates with heterochromatin and integral inner nuclear membrane proteins providing links among the genome nucleoskeleton and cytoskeleton. The lamins are type v intermediate filaments which can be categorized as either a type lamin a c or b type lamin b 1 b 2 according to homology of their dna sequences biochemical properties and cellular localization during the cell cycle.
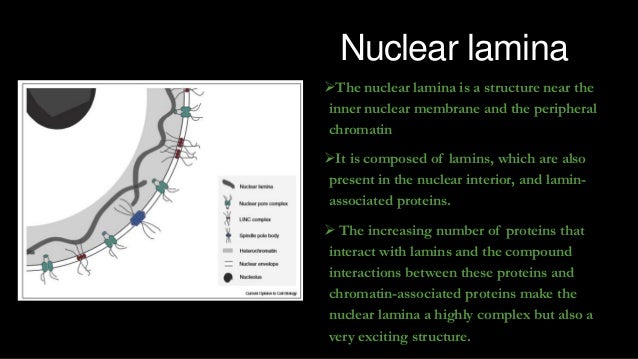
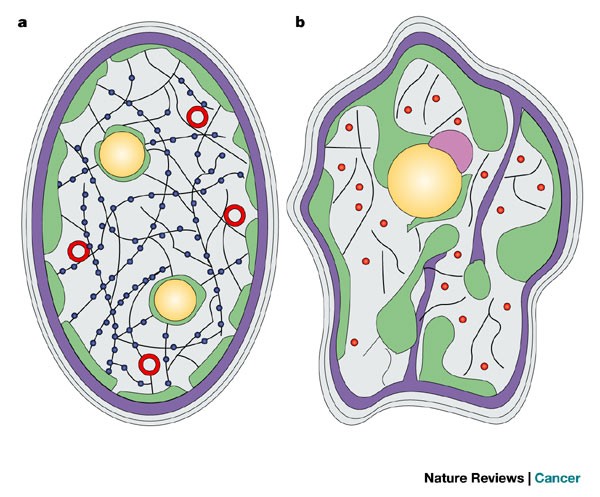
Lamin a is also farnesylated but it is unique among farnesylated human proteins 90. The increasing number of proteins that interact with lamins and the compound interactions between t. This protein lattice associates with heterochromatin and integral inner nuclear membrane proteins providing links among the genome nucleoskeleton and cytoskeleton. Lamins have elastic and mechanosensitive properties and can alter gene regulation in a feedback response to mechanical cues.
Nuclear lamins interact with inner nuclear membrane proteins to form the nuclear lamina on the interior of the nuclear envelope. The nuclear lamina is a multi protein lattice composed of a and b type lamins and their associated proteins. Following modifications by ftase rce1 and icmt prelamin a undergoes a farnesylation dependent proteolytic cleavage catalyzed by the zinc metalloprotease zmpste24. It is composed of lamins which are also present in the nuclear interior and lamin associated proteins.
Lamins are present in all animals but are not found in microorganisms. The nuclear lamina consists of lamin proteins that are required for nuclear envelope assembly 87 88. In the 1990s mutations.