Thiol Vinyl Ether

Vinyl ether functionalized cyclic phosphotriester monomer 2 ethylene glycol vinyl ether 1 3 2 dioxaphospholane 2 oxide evep 1 was synthesized by following a typical condensation method.
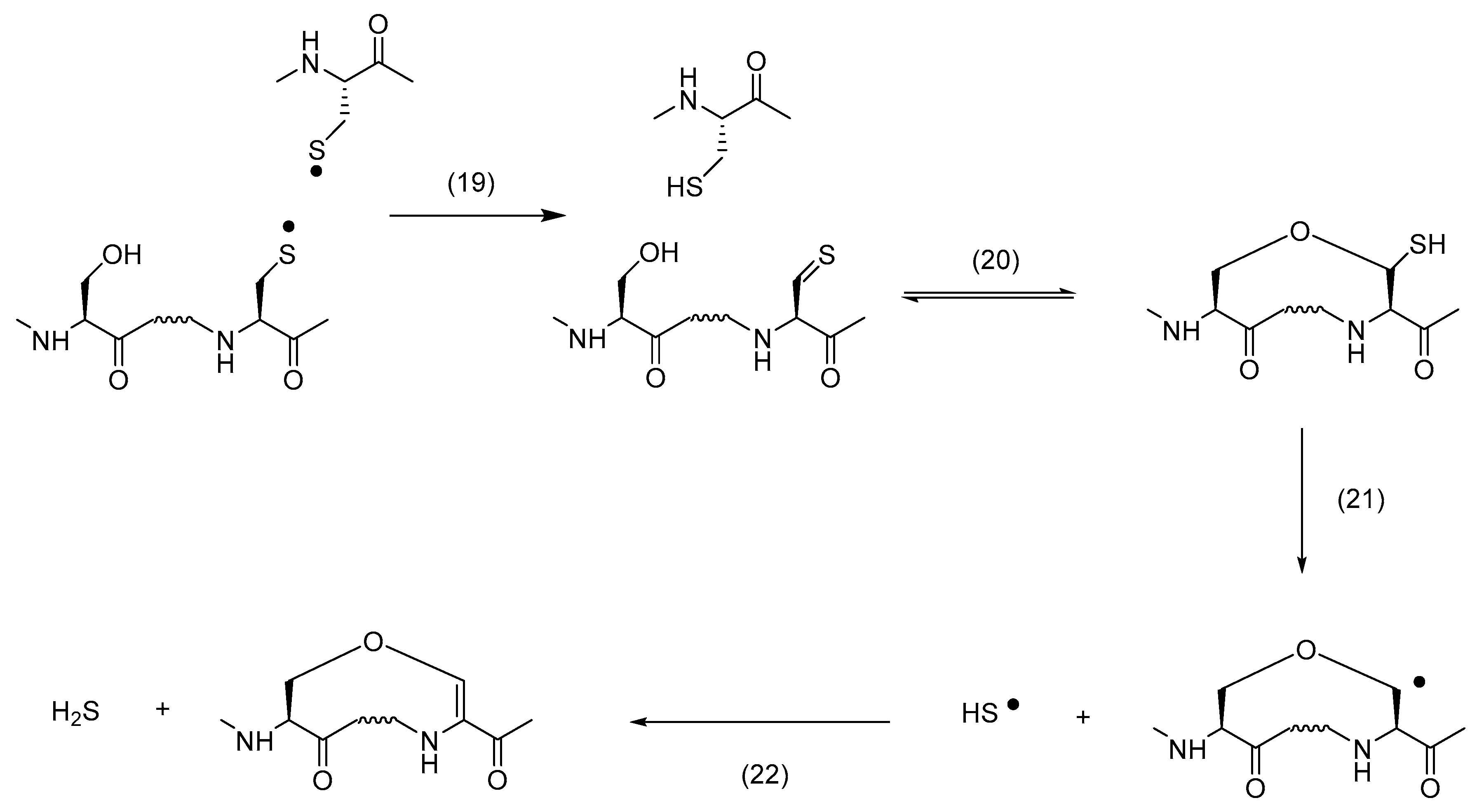
Thiol vinyl ether. P toluenesulfonic acid ptsa induced a cationic thiol ene reaction to generate a thioacetal in high yield whereas 2 2 azobisisobutyronitrile resulted in a radical thiol ene reaction to give a thioether also in high yield. Ethyl isobutyl ether allyl vinyl ether. These thiol ene polymerizations are photoinitiated without the use of photoinitiator molecules. The sequential thiol vinyl ether copolymerization and the vinyl ether cationic polymerization result in cross linked networks with thermal and mechanical properties that are combinations of each system.
Structure and properties of ethers the geometry around the o atom of an ether ror is similar to that. While thiol ene reactions of vinyl ethers with various thiols yield robust thioether linkages viareaction at the β carbon reactions with thiols or alcohols in the presence of acid. Thiol ene free radical and vinyl ether cationic hybrid photopolymerization macromolecules. 1 product result match criteria.
A thiol monomer is shown to copolymerize with vinyl ether allyl acrylate methacrylate and vinylbenzene monomers. C 3 h 6 o n. Poly methyl vinyl ether solution. The advantages of the vinyl ether functionality is its capability to form stable or alternatively dynamic covalent bonds.
The kinetics of the vinyl and upe group reactions are mirrored by the thiol conversion in figure 12 a which exhibits a very rapid initial loss of thiol groups reaction with vinyl ether followed by a much slower conversion reaction with upe. This reaction was first reported in 1905 but it gained prominence in the late 1990s and early 2000s for its feasibility and wide range of applications. Methoxy peg thiol methoxypolyethylene glycol thiol. Coupling of ethylene glycol vinyl ether eve to 2 chloro 3 oxo 1 3 2 dioxaphospholane cop in the presence of triethylamine tea in dichloromethane dcm.
Product name linear formula. Thiol ene cationic and radical reactions were conducted for 1 1 addition between a thiol and vinyl ether and also for cyclization and step growth polymerization between a dithiol and divinyl ether.